On November 12, 2024, the Department of Radiotherapy of Sun Yat-sen University Cancer Prevention and Treatment Center (SUNYCCPC) successfully completed the first patient enrollment in the "KSD-101 Early Exploratory Clinical Study in EBV-associated Nasopharyngeal Carcinoma that Failed Standard Treatment" project.
▲Return site
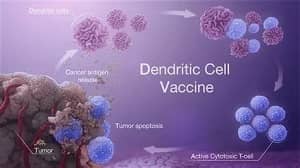
KSD-101 is an autologous dendritic cell vaccine (DC vaccine) independently developed by Hengsai Biotechnology, loaded with EBV-associated tumor-like complex antigens, which can activate EBV-specific CTLQ in the human body after subcutaneous injection, thus realizing the effective recognition and killing of tumor cells.KSD-101 has been successfully completed the dual reporting of the U.S.-China, and has been granted IND authorization by U.S. Food and Drug Administration. KSD-101 has successfully completed both Chinese and American reports, and has been granted IND license by the U.S. Food and Drug Administration (FDA), which is the first original dendritic cell vaccine (DC vaccine) product in China that has been granted IND license by the U.S. FDA.
The safety and efficacy of KSD-101 has been preliminarily validated in clinical IIT studies for the treatment of relapsed and refractory hematological tumors, and a multicenter clinical IIT study for nasopharyngeal carcinoma is currently underway at Tongji Hospital of Tongji Medical College, Huazhong University of Science and Technology, and at Sun Yat-sen University Cancer Prevention and Control Center.
 Hengsai Biotechnology
Hengsai Biotechnology